You are here
Content
Translational pain research
In this research group sensitization of primary afferent nociceptors is investigated as one mechanism for inflammatory and neuropathic chronic pain. We focus on translational studies using single fiber recording techniques in chronic pain patients, human volunteers and large animals (pig) combined with functional and structural investigations of skin innervation in-vivo and in cell-culture.
- Cellular porcine model
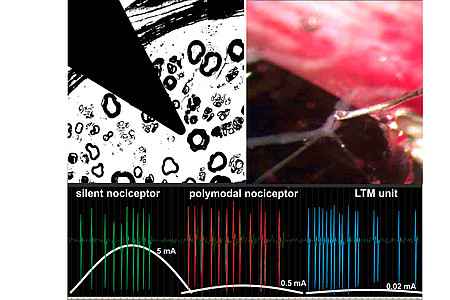
Patch-clamp and histochemistry of porcine dorsol root ganglion cells to characterize different nociceptor classes. Development of in-vitro model of nerve endings separated from the cell body and coculture with keratinocytes. - Porcine single fiber recordings
Characterization of different afferent nerve fiber classes and modulation of axonal excitability using standard teased fiber techniques in the pig saphenous nerve. Effects of locally applied growth factors on excitability of nerve endings and axonal excitability. - Porcine neurogenic inflammation
Development of experimental models of peripheral sensitization by UVB irradiation with the objective readout of axon reflex erythema measured by laser Doppler imaging. Microdialysis in the sunburn to assess levels of inflammatory mediators involved in peripheral sensitization. - Human pain models
Characterization of human pain models for peripheral (UVB irradiation) and central (electrical pain and hyperalgesia model) sensitization by pharmacological intervention. Objective assessment of cutaneous unmyelinated innervation by electrically induced axon reflex erythema and electrically induced axon reflex sweating. - Chronic pain patients
Microneurographic characterization of neuropathic changes in primary afferent fibers linked to chronic pain. Correlation of functional and structural changes in neuropathic pain by using skin biopsies, quantitative sensory testing, and electrically induced axon reflex measurements.
National and International Joint Research Programs
- Research Group FOR 2690: "Translational Pruritus Research" (Speaker)
- Heidelberg Pain Consortium SFB 1158: Project A01: "Structure-function properties and markers of silent nociceptors in mice and humans" (Member)
- International Research Training Group GRK 1874 DIAMICOM (Diabetic Microvascular Complications)(Member)
Project MD SP14: "Diabetic thin fiber neuropathy – objective assessment and mechanism"
Selection of recent publications
- Gonzalez-Rodriguez S, Quadir MA, Gupta S, Walker KA, Zhang X, Spahn V, Labuz D, Rodriguez-Gaztelumendi A, Schmelz M, Joseph J, Parr MK, Machelska H, Haag R, Stein C. Polyglycerol-opioid conjugate produces analgesia devoid of side effects. Elife 2017;6.
- Jonas R, Klusch A, Schmelz M, Petersen M, Carr RW. Assessment of TTX-s and TTX-r Action Potential Conduction along Neurites of NGF and GDNF Cultured Porcine DRG Somata. PLoS ONE 2015;10(9):e0139107.
- Jonas R, Namer B, Stockinger L, Chisholm K, Schnakenberg M, Landmann G, Kucharczyk M, Konrad C, Schmidt R, Carr R, McMahon S, Schmelz M, Rukwied R. Tuning in C-nociceptors to reveal mechanisms in chronic neuropathic pain. Ann Neurol 2018;83(5):945-957.
- Jonas R, Prato V, Lechner SG, Groen G, Obreja O, Werland F, Rukwied R, Klusch A, Petersen M, Carr RW, Schmelz M. TTX-Resistant Sodium Channels Functionally Separate Silent From Polymodal C-nociceptors. Frontiers in Cellular Neuroscience 2020;14(13).
- Klusch A, Gorzelanny C, Reeh PW, Schmelz M, Petersen M, Sauer SK. Local NGF and GDNF levels modulate morphology and function of porcine DRG neurites, In Vitro. PLoS ONE 2018;13(9):e0203215.
- Namer B, Orstavik K, Schmidt R, Kleggetveit IP, Weidner C, Mork C, Kvernebo MS, Kvernebo K, Salter H, Carr TH, Segerdahl M, Quiding H, Waxman SG, Handwerker HO, Torebjork HE, Jorum E, Schmelz M. Specific changes in conduction velocity recovery cycles of single nociceptors in a patient with erythromelalgia with the I848T gain-of-function mutation of Nav1.7. Pain 2015;156(9):1637-1646.
- Obreja O, Rukwied R, Nagler L, Schmidt M, Schmelz M, Namer B. Nerve growth factor locally sensitizes nociceptors in human skin. Pain 2018;159(3):416-426.
- Sauerstein K, Liebelt J, Namer B, Schmidt R, Rukwied R, Schmelz M. Low-Frequency Stimulation of Silent Nociceptors Induces Secondary Mechanical Hyperalgesia in Human Skin. Neuroscience 2018;387:4-12.
- Steinhoff M, Schmelz M, Szabo IL, Oaklander AL. Clinical presentation, management, and pathophysiology of neuropathic itch. Lancet Neurol 2018;17(8):709-720.
- Yosipovitch G, Greaves MW, Schmelz M. Itch. The Lancet 2003;361(9358):690-694.
Context Column
Contact

Prof. Dr. Martin Schmelz
Experimental Pain Research
Medical Faculty Mannheim
Ludolf-Krehl-Str. 13-17
68167 Mannheim
Phone +49 621 383-71650
martin.schmelz@medma.uni-heidelberg.de
