You are here
Content
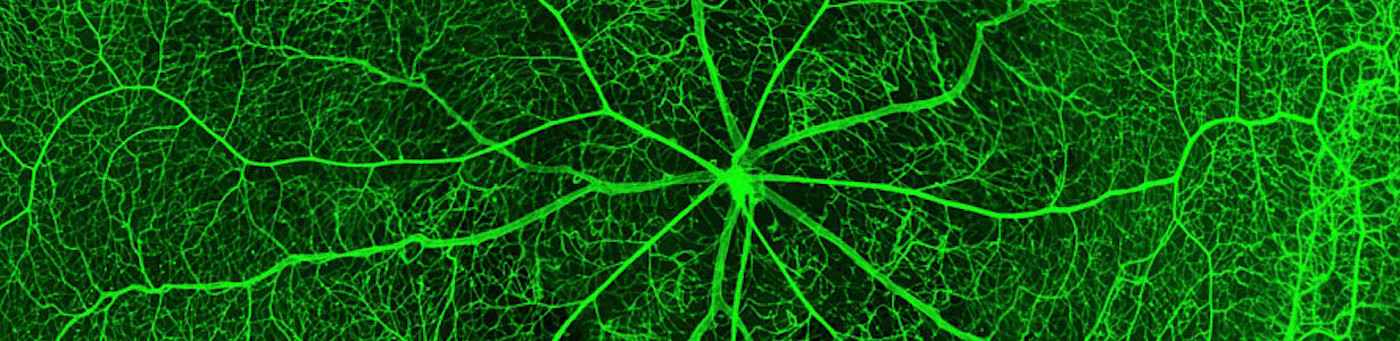
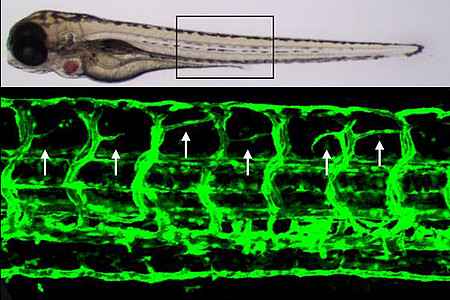
During the last decades the development and function have blood vessels has been intensively studied and several fundamental biochemical, molecular and cellular mechanisms have been identified. In addition to several established cell culture systems, animal model systems including mice, rat and zebrafish, have served as a basis to understand fundamental processes of vascular development and vascular function in vivo. Yet, mechanisms leading to a diseased vasculature under pathological conditions, such as in diabetes mellitus, and how these alterations contribute to development of diabetic late complications are less understood. Thus, our work aims to identify and characterize diabetes induced vascular complications in zebrafish.
Specifically, we will address the following questions:
- Which signaling cascades are altered by hyperglycemia and by metabolic intermediates (reactive metabolites) in zebrafish?
- Which cellular and molecular alterations are induced by hyperglycemia and by reactive metabolites and how contribute these alterations to dysfunctional blood vessels, kidneys and neurons in zebrafish?
- How can the identified diabetes-induced alterations in zebrafish be prevented or regressed?
- Which significances have the identified alterations in zebrafish in the development of diabetic late complications (diabetic retinopathy, nephropathy and neuropathy) in human?
Project-related publications
Context Column
Kontakt

Prof. Dr. Jens Kroll
Working Group Vascular Signaling
Department of Vascular Biology and Tumor Angiogenesis
Center for Biomedicine and Medical Technology Mannheim (CBTM)
Medical Faculty Mannheim
Heidelberg University
Ludolf-Krehl-Str. 13-17
68167 Mannheim
Phone +49 621 383-9965
jens.kroll@medma.uni-heidelberg.de