You are here
Content
Our research interests focus on regenerative medicine, especially the development of novel cell based therapies (advanced therapy medicinal products – ATMP). Within the last years this field has been marked by significant achievements, further boosted by awarding the two stem cell researchers S. Yamanaka and J. Gurdon with the 2012 Nobel Prize in Medicine.
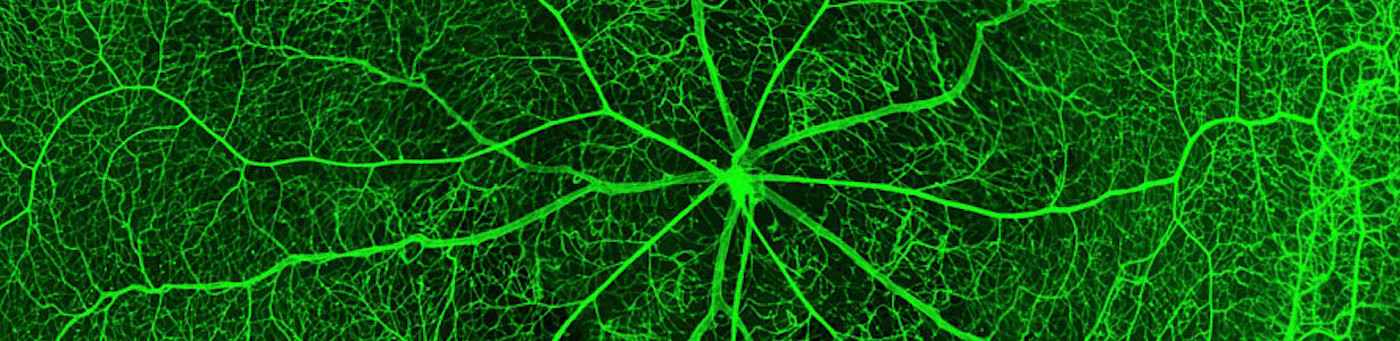
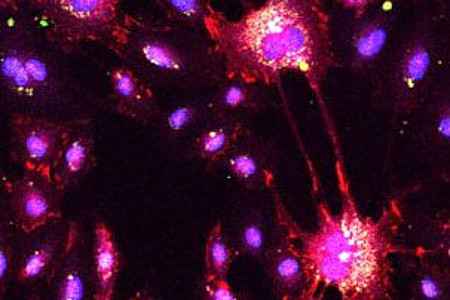
We are in particular fascinated by two cell populations due to their promising wide therapeutic spectra in vascular and regenerative medicine: mesenchymal stromal cells (MSC) and endothelial progenitor cells (EPC).
Our special interests are:
- to understand the source and the regenerative properties of these cells
- to optimize their proliferative and functional capacities focusing on potential therapeutic targets in vascular and regenerative medicine
- to identify and optimize the mechanisms of action, especially immunomodulatory and pro-regenerative factors
- to better understand homing and engraftment to be able to modulate these processes
- to manufacture cells for clinical studies in vascular medicine according to good manufacturing practice (GMP) guidelines
Beyond this we are running the Core Facility for Flow Cytometry and Cell Sorting, initiated in 2007 by a HBFG grant. The facility belongs to the Centre for Biomedicine and Medical Technology (CBTM) and is open to researchers to perform cell sorting and flow cytometry experiments.
Project-related publications
- Kremer H, Gebauer J, Elvers-Hornung S, Uhlig S, Hammes HP, Beltramo E, Steeb L, Harmsen MC, Sticht C, Klueter H, Bieback K, Fiori A. Pro-angiogenic Activity Discriminates Human Adipose-Derived Stromal Cells From Retinal Pericytes: Considerations for Cell-Based Therapy of Diabetic Retinopathy.Front Cell Dev Biol. 2020;8:387. doi: 10.3389/fcell.2020.00387.
- Fiori A, Hammes HP, Bieback K. Adipose-derived mesenchymal stromal cells reverse high glucose-induced reduction of angiogenesis in human retinal microvascular endothelial cells. Cytotherapy. 2020;22(5):261-275. doi: 10.1016/j.jcyt.2020.02.005.
- Torres Crigna A, Fricke F, Nitschke K, Worst T, Erb U, Karremann M, Buschmann D, Elvers-Hornung S, Tucher C, Schiller M, Hausser I, Gebert J, Bieback K. Inter-Laboratory Comparison of Extracellular Vesicle Isolation Based on Ultracentrifugation. Transfus Med Hemother. doi:10.1159/000508712)
- Worst TS, Previti C, Nitschke K, Diessl N, Gross JC, Hoffmann L, Frey L, Thomas V, Kahlert C, Bieback K, Crigna AT, Fricke F, Porubsky S, Westhoff N, Hardenberg JV, Nuhn P, Erben P, Michel MS, Boutros M. miR-10a-5p and miR-29b-3p as Extracellular Vesicle-Associated Prostate Cancer Detection Markers. Cancers (Basel). 2019 Dec 21;12(1):43. doi: 10.3390/cancers12010043.
- Czabanka M, Petrilli LL, Elvers-Hornung S, Bieback K, Albert Imhof B, Vajkoczy P, Vinci M. Junctional Adhesion Molecule-C Mediates the Recruitment of Embryonic-Endothelial Progenitor Cells to the Perivascular Niche during Tumor Angiogenesis.Int J Mol Sci. 2020 Feb 11;21(4):1209. doi: 10.3390/ijms21041209.
- Netsch P, Elvers-Hornung S, Uhlig S, Klüter H, Huck V, Kirschhöfer F, Brenner-Weiß G, Janetzko K, Solz H, Wuchter P, Bugert P, Bieback K. Human mesenchymal stromal cells inhibit platelet activation and aggregation involving CD73-converted adenosine. Stem Cell Res Ther. 2018 Jul 4;9(1):184. doi: 10.1186/s13287-018-0936-8.
- Fiori A, Terlizzi V, Kremer H, Gebauer J, Hammes HP, Harmsen MC, Bieback K. Mesenchymal stromal/stem cells as potential therapy in diabetic retinopathy.Immunobiology. 2018 Dec;223(12):729-743. doi: 10.1016/j.imbio.2018.01.001.
- Huang H, Kolibabka M, Eshwaran R, Chatterjee A, Schlotterer A, Willer H, Bieback K, Hammes HP, Feng Y.Intravitreal injection of mesenchymal stem cells evokes retinal vascular damage in rats. FASEB J. 2019 Dec;33(12):14668-14679. doi: 10.1096/fj.201901500R.
- Grudzenski S, Baier S, Ebert A, Pullens P, Lemke A, Bieback K, Dijkhuizen RM, Schad LR, Alonso A, Hennerici MG, Fatar M. The effect of adipose tissue-derived stem cells in a middle cerebral artery occlusion stroke model depends on their engraftment rate. Stem Cell Res Ther. 2017 Apr 26;8(1):96. doi: 10.1186/s13287-017-0545-y.
- Sharma KR, Heckler K, Stoll SJ, Hillebrands JL, Kynast K, Herpel E, Porubsky S, Elger M, Hadaschik B, Bieback K, Hammes HP, Nawroth PP, Kroll J. ELMO1 protects renal structure and ultrafiltration in kidney development and under diabetic conditions. Sci Rep. 2016 Nov 16;6:37172. doi: 10.1038/srep37172.
Context Column
Contact

Karen Bieback, PhD
Institute of Transfusion Medicine and Immunology
Medical Faculty Mannheim
Universität Heidelberg
German Red Cross Blood Service Baden-Württemberg - Hessen
Friedrich-Ebert-Str. 107
68167 Mannheim
Phone +49 621 383-9720
karen.bieback@medma.uni-heidelberg.de