You are here
Content
We are working on several aspects of improved oncological radiotherapy treatment planning and monitoring by using physiological and functional imaging of CT, MRI and PET. One of our main research aspects lies in developing new MR techniques (23Na imaging, dynamic MRT, diffusion, perfusion, blood bolus tagging, BOLD MRI) for clinical use in therapy planning and monitoring. Another main aspect denotes imaging of hyperpolarized 3He in the human lung, as well as T2*- and T1-techniques for non-invasive measurement of tissue oxygenation and perfusion in the myocardium which is of general interest in radiology.
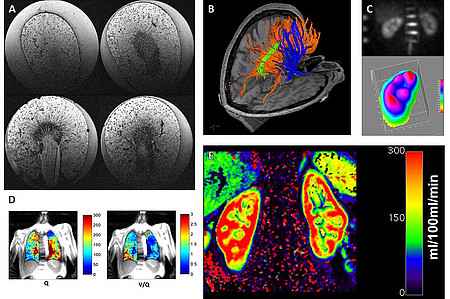
Our overall goal is the evaluation of quantitative biomarkers for diagnosis and/or therapy monitoring accomplished by translational animal/cell experiments. Major contributions are the evaluation of glomeruli counts in rat kidneys using MR at 9.4 T (Figure 1A); the establishment of a high-resolution protocol for diffusion tensor imaging at 3 T for tracking fibers in the brain and the clinical evaluation of diffusion kurtosis in whole body applications (Figure 1B); the development of an MR set up for measuring sodium concentration in human kidneys at 3 T (Figure 1C); the implementation of a protocol for use in clinical contexts for mapping of perfusion and ventilation in the human lung at 1.5 T (Figure 1D); and the development of an MR set up to measure perfusion maps in the human kidney for quantitative evaluation at 3 T (Figure 1E).
We do have many years of experi¬ence (since 1985) in developing new methods and techniques for magnetic reso¬nance imaging with strong focus on Medical Technology using MR imaging and spectroscopy for modern treatment planning and monitoring. The institute is equipped with 3x 1.5T and 2x 3T whole-body MR systems and has also access to a 9.4T animal system at the Central Institute of Mental Health. Besides, we are developing molecular innovative imaging technologies by fusion of several imaging modalities (CT, MRI, PET) to enable image-guided, high-precision interventions using high-end CT and robotic systems (ZEEGO, Siemens). The group is composed of scien-tists from physics, electrical engineering, and computer science and is working in close co-operation with the local medical departments (Radiology, Radiotherapy, Neuroradiology, Neurology and Cardiology). The aim of the research is to design innovative diagnostic methods and procedures to optimize morphologi¬cal, functional and metabolic diagnosis as a basis for radiology and image-guided radiotherapy, as well as to establish an experimental clinical translational research using high-resolution animal imaging. The focus of the current development work are methods for physiological MR imaging (perfusion, diffusion, oxygenation) on hu¬mans especially in the abdomen (kidney, lung, heart), as well as sodium imaging for non-invasive measurement of tissue viability. A further objective is the establishment of animal models (rat and mouse) for building various translational studies (high-resolution morphology, X-nuclear imaging, stroke model and renal glomeruli representa¬tion) accomplished by cell experiments in a bioreactor system.
National and International Joint Research Projects
- BMBF M2OLIE
- DFG-Radiomics ZO 257/8-1
- DFG-FDLunge ZO 257/6-1
- DFG-Radiomics RectumCA ZO 257/9-1
Selected publications
Bertleff M, Domsch S, Weingärtner S, Zapp J, O'Brien K, Barth M, Schad LR. Diffusion parameter mapping with the combined intravoxel incoherent motion and kurtosis model using artificial neural networks at 3 T. NMR Biomed 2017;30(12):e3833.
Weingärtner S, Meßner NM, Zöllner FG, Akçakaya M, Schad LR. Black-blood native T1 mapping: Blood signal suppression for reduced partial voluming in the myocardium. Magn Reson Med 2017;78(2):484-493.
Rieger B, Akçakaya M, Pariente JC, Llufriu S, Martinez-Heras E, Weingärtner S, Schad LR. Time efficient whole-brain coverage with MR Fingerprinting using sliceinterleaved echo-planar-imaging. Sci Rep 2018;8:6667.
Neumann W, Lehnart V, Vetter Y, Bichert A, Schad LR, Zöllner FG. Coupled actuators with a mechanically synchronized phase during MR elastography: A phantom feasibility study. Concepts Magn Reson Part B 2018;48B:e21403.
Chung K, Schad LR, Zöllner FG. Tomosynthesis implementation with adaptive online calibration on clinical C-arm systems. Int J Comput Assist Radiol Surg 2018;13:1481-1495.
Davids M, Guérin B, vom Endt A, Schad LR, Wald LL. Prediction of peripheral nerve stimulation thresholds of MRI gradient coils using coupled electromagnetic and neurodynamic simulations. Magn Reson Med 2019;81(1):686-701.
Malzacher M, Chacon-Caldera J, Paschke N, Schad LR. Feasibility study of a double resonant 8-channel 1H/ 8-channel 23Na receive-only head coil at 3 Tesla. Magn Reson Imaging 2019;59:97-104.
Hoesl MAU, Kleimaier D, Hu R, Malzacher M, Nies C, Gottwald E, Schad LR. Na Triple-quantum signal of in vitro human liver cells, liposomes, and nanoparticles: Cell viability assessment vs. separation of intra- and extracellular signal. J Magn Reson Imaging 2019;50(2):435-444.
Hubertus S, Thomas S, Cho J, Zhang S, Wang Y, Schad LR. Using an artificial neural network for fast mapping of the oxygen extraction fraction with combined QSM and quantitative BOLD. Magn Reson Med 2019;82(6):2199-2211.
Ruttorf M, Kristensen S, Schad LR, Almeida J. Transcranial Direct Current Stimulation Alters Functional Network Structure in Humans: A Graph Theoretical Analysis. IEEE Trans Med Imaging 2019;38(12):2829-2837.
Context Column
Contact

Prof. Dr. Lothar R. Schad
Computer Assisted Clinical Medicine
Medical Faculty Mannheim
Ludolf-Krehl-Straße 13-17
68167 Mannheim
Phone +49 621 383-5123
lothar.schad@medma.uni-heidelberg.de
