Sie befinden sich hier
Inhalt
30th March 2023
Multiple sclerosis (MS) is the most common chronic inflammatory disease of the central nervous system (CNS). It is characterized by demyelination, causing damage to the protective myelin sheath that surrounds nerve fibers and, as the disease progresses, destruction of neurons. In an international study, scientists from the Department of Neurology at the University Medical Center Mannheim (UMM), in collaboration with colleagues from Düsseldorf, Münster, Cambridge and San Francisco, have now identified potassium channels along the nerve fibers in the CNS as potential targets to arm vulnerable neurons against the inflammatory demyelination that occurs in the course of MS.
Underlying the new strategy is an intriguing, but so far unproven, hypothesis that a major factor in nerve damage in MS is chronic neuronal hyperexcitability: In animal models of MS and other in vivo models of de- and remyelination, evidence is accumulating that susceptible neurons perish over time due to metabolic exhaustion caused by chronic hyperexcitability. Normalizing the excitability of neurons therefore appears to be a promising strategy to prevent neurodegeneration.
The increased excitability of neurons is likely the result of several factors that lower the threshold for action potential generation associated with chronic inflammatory demyelination. Normalizing ion imbalance could therefore be an interesting therapeutic target in MS.
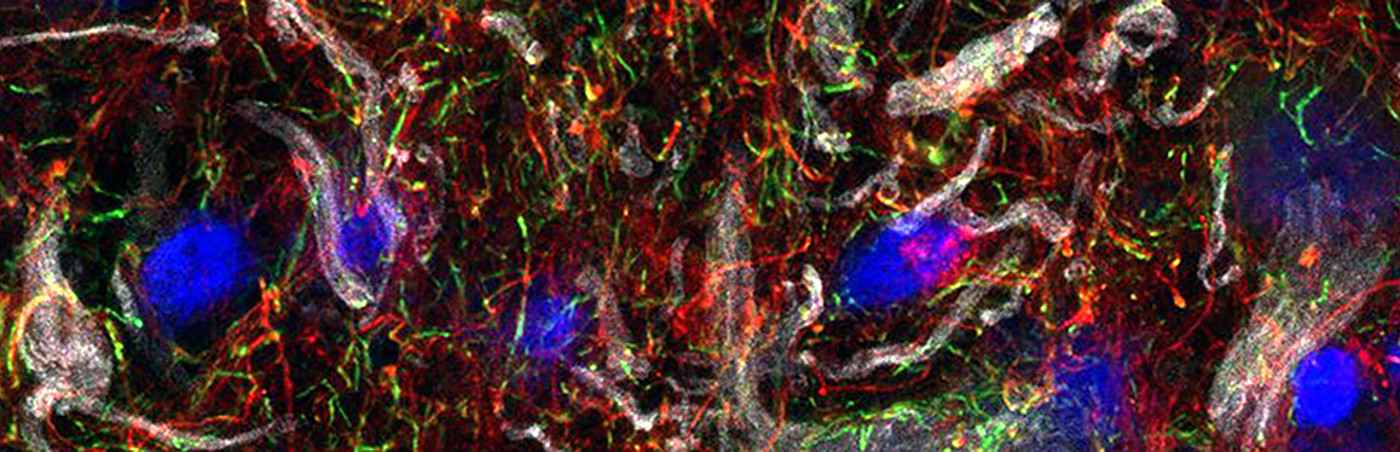
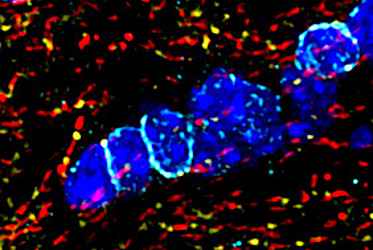
The myelin layer lining the axons of nerve cells is interrupted at regular intervals by so-called nodes of Ranvier also known as myelin-sheath gaps. They serve to speed up transmission of electrical excitations. Since potassium channels play an important role in regulating the excitability of nerve cells at and around nodes of Ranvier, the scientists took a closer look at the corresponding channels: Outwardly rectifying potassium channels (Kv channels) of axons, permeable to outward flow of ions, and inwardly rectifying potassium channels (Kir channels) of oligodendrocytes, which form the myelin layer of neurons and conduct potassium ions predominantly from extracellular to intracellular.
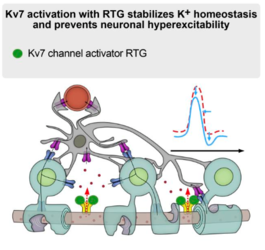
Studies on the spatial and functional relationship between Kv7 channels and Kir4.1 channels in the healthy state and under inflammatory-demyelinating conditions demonstrated that the regulation of both channels is disturbed in MS and in experimental animal models. A positive effect was shown by the administration of retigabine, a compound that specifically opens Kv7 channels. Originally developed for epilepsy, retigabine reduced neuronal hyperexcitability in humans and animals and improved clinical symptoms, at least in animal models.
"We have thus identified a new therapeutic approach to pharmacologically protect nerve cells during MS disease development," says Professor Lucas Schirmer, MD (University Medical Center Mannheim, Germany), who is one of the corresponding authors of the current study, along with Professor Sven Meuth, MD (University Hospital Düsseldorf, Germany) and Professor David Rowitch, MD (University of Cambridge).
The scientists' results suggest that compensatory interactions between neurons and oligodendrocytes through Kv7 and Kir4.1 channels promote resilience of damaged neurons and that drug activation of Kv7 channels could be a promising strategy to protect neurons from inflammatory demyelination.
Original scientific publication:
Neuron-oligodendrocyte potassium shuttling at nodes of Ranvier protects against inflammatory demyelination
2023 Journal of Clinical Investigation (JCI), Volume 133, issue 7
DOI: https://doi.org/10.1172/JCI164223
Press release at the Medical Faculty Mannheim (German language):
30.03.2023 Neues aus der MS-Forschung: Kaliumkanäle als Strategie, um Nervenzellen und Entmarkung zu schützen
Contact for scientific information:
Prof. Dr. med. Lucas Schirmer
Heisenberg Professor for Translational Neurobiology
Department of Neurology | University Medical Center Mannheim
Medical Faculty Mannheim of Heidelberg University
Theodor-Kutzer-Ufer 1-3 | 68167 Mannheim
lucas.schirmer@medma.uni-heidelberg.de