Sie befinden sich hier
Inhalt
13th February 2023
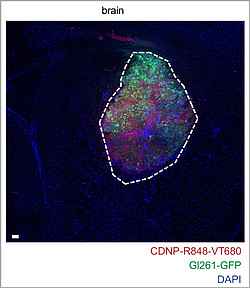
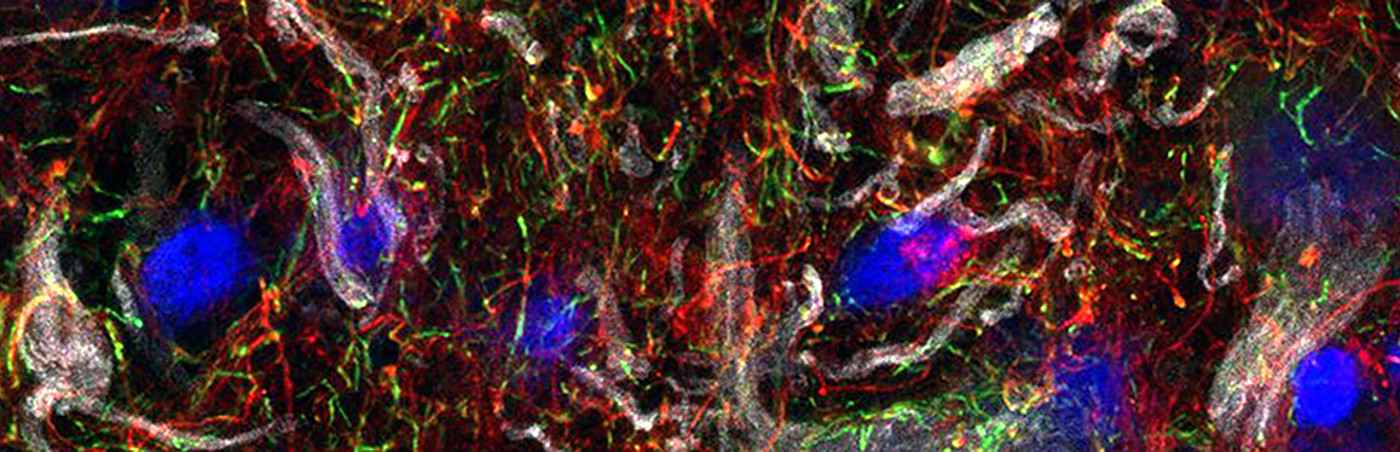
Glioblastoma, the most common and aggressive primary brain tumor type, is considered an immunologically “cold” tumor with sparse infiltration by adaptive immune cells. Immunosuppressive tumor-associated myeloid cells are drivers of tumor progression. Therefore, targeting and reprogramming intratumoral myeloid cells is an appealing therapeutic strategy. Michael Platten and his team investigated a β-cyclodextrin nanoparticle (CDNP) formulation encapsulating the Toll-like receptor 7 and 8 (TLR7/8) agonist R848 (CDNP-R848) to reprogram myeloid cells in the glioma microenvironment. The research team showed that intravenous monotherapy with CDNP-R848 induces regression of established syngeneic experimental glioma, resulting in increased survival rates. Mechanistically, CDNP-R848 treatment reshapes the immunosuppressive tumor microenvironment and orchestrates tumor clearing by pro-inflammatory tumor-associated myeloid cells, independently of immune cells, such as T cells and natural killer cells. In conclusion, CDNP-R848 induces tumor regression in experimental glioma by targeting blood-borne macrophages without requiring adaptive immunity. This underlines that the suppressive myeloid compartment in glioma can be specifically targeted, thus paving the way for new, druggable targets for glioma immunotherapy that can be assessed by phagocyte-directed imaging. (modified abstract from Turco et al., Nat Commun, 2023)
Original scientific publication:
T cell-independent eradication of experimental glioma by intravenous TLR7/8-agonist-loaded nanoparticles
Turco V, Pfleiderer K, Hunger J, Horvat NK, Karimian-Jazi K, Schregel K, Fischer M, Brugnara G, Jähne K, Sturm V, Streibel Y, Nguyen D, Altamura S, Agardy DA, Soni SS, Alsasa A, Bunse T, Schlesner M, Muckenthaler MU, Weissleder R, Wick W, Heiland S, Vollmuth P, Bendszus M, Rodell CB, Breckwoldt MO, Platten M.
2023, Nat Commun 14(1):771
DOI: https://doi.org/10.1038/s41467-023-36321-6